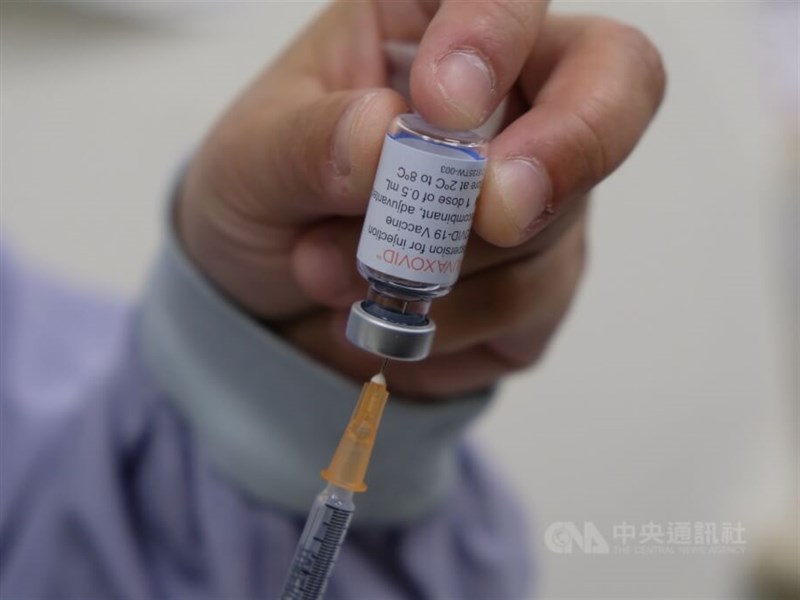
Taiwan's Food and Drug Administration (FDA) has granted emergency use authorization (EUA) for Moderna's Spikevax XBB.1.5 COVID-19 vaccine and is planning to roll out the new jab by the end of this month.
(Full text of the story is now in CNA English news archive. To view the full story, you will need to be a subscribed member of the CNA archive. To subscribe, please read here.)
More in CORONAVIRUS
-
![Taiwan to shorten COVID-19 vaccine interval to 2 months for high-risk groups]() Taiwan to shorten COVID-19 vaccine interval to 2 months for high-risk groupsThe Centers for Disease Control (CDC) announced Thursday that the required interval between two COVID-19 vaccine doses for high-risk groups will be shortened from six months to two months, effective next Wednesday.06/06/2025 08:17 PM
Taiwan to shorten COVID-19 vaccine interval to 2 months for high-risk groupsThe Centers for Disease Control (CDC) announced Thursday that the required interval between two COVID-19 vaccine doses for high-risk groups will be shortened from six months to two months, effective next Wednesday.06/06/2025 08:17 PM -
![CDC confirms ample supply of COVID-19 drugs, vaccines]() CDC confirms ample supply of COVID-19 drugs, vaccinesThe Centers for Disease Control (CDC) reiterated Sunday that Taiwan has sufficient antiviral medication for COVID-19 patients and emphasized the importance of vaccination, amid concerns raised by public figures.06/01/2025 06:13 PM
CDC confirms ample supply of COVID-19 drugs, vaccinesThe Centers for Disease Control (CDC) reiterated Sunday that Taiwan has sufficient antiviral medication for COVID-19 patients and emphasized the importance of vaccination, amid concerns raised by public figures.06/01/2025 06:13 PM -
![CDC issues COVID-19 warning as Dragon Boat Festival long weekend begins]() CDC issues COVID-19 warning as Dragon Boat Festival long weekend beginsMembers of the public should not let their guard down on COVID-19 and seek medical help if they experience symptoms that indicate risk of severe infection, the Centers for Disease Control (CDC) said Friday, the first day of the Dragon Boat Festival long weekend in Taiwan.05/30/2025 05:04 PM
CDC issues COVID-19 warning as Dragon Boat Festival long weekend beginsMembers of the public should not let their guard down on COVID-19 and seek medical help if they experience symptoms that indicate risk of severe infection, the Centers for Disease Control (CDC) said Friday, the first day of the Dragon Boat Festival long weekend in Taiwan.05/30/2025 05:04 PM
Latest
-
Politics
U.S. envoy calls on Taiwan legislators to pass special defense budget
03/26/2026 09:12 PM -
Politics
Clashes erupt outside Taipei court as Ko gets 17 years
03/26/2026 09:01 PM -
Business
Unemployment rate rises to 3.32% in February, ending 5-month decline
03/26/2026 07:48 PM -
Politics
Ko faces 2028 presidential race ban after receiving 17-year sentence
03/26/2026 07:05 PM -
Politics
Key developments in cases involving ex-Taipei mayor Ko Wen-je
03/26/2026 04:52 PM